Despite the fact that many patients still consider that all drugs are incredibly harmful and may cause toxic effects on different organs, it is not so at all. All drugs before they get to the pharmacy are carefully checked out and studied to exclude their dangerous effect on human body. Of course, there is a list of the drugs having rather serious adverse effects, but the indications for their use are strictly limited, and these drugs are used only in case of very serious diseases.
Struggle for reproductive health
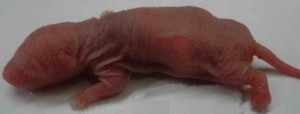
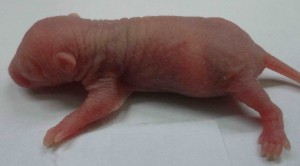
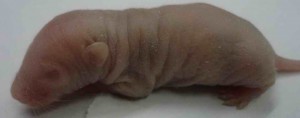
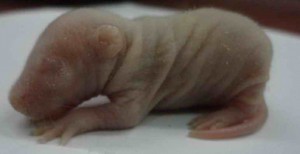
One of the important aspects falling under attention of such preclinical studies is a reproductive toxicity of the drug. This term means an ability of a substance to cause toxic effect on reproductive organs of a man or a woman followed by a decrease in sexual function and ability to reproduce, and to exert pathological influence on development of the offspring.
All necessary preclinical studies on reproductive toxicity of the drug are carried out to exclude such adverse pathologies. For these experiments different animals are used, such as:
- mice;
- rats;
- rabbits;
- guinea pigs and others.
This research work allows determining reproductive toxicity of the drug that in the future will give a chance to prevent infertility development in patients and negative influence on a fetus followed by an impact on a newborn child in case of drug administration throughout pregnancy. If reproductive toxicity of a substance exceeds acceptable standards, this drug will be sent to improve or be taken out of production on the stage of preclinical study. Also it is possible to search such dose of the drug that retains the desired therapeutic effect, and its reproductive toxicity remains at an acceptable level.
Reproductive toxicity study of drugs is performed according to the Manual for conducting preclinical studies of drugs. Under the editorship of Mironov A.N., Bunatyan N.D. et al., M., publishing house “Grif and K”, 2012, FDA – Integration of Study Results to Assess Concerns about Human Reproductive and Developmental Toxicities, ICH – S5(R2).
Assessment of toxic effects on reproductive system, the sudy of embrio-and fetotoxicity by indicators such as preimplantation and post-implantation mortality with an assessment of teratogenicity. The parameters assessed are the follows:
- number of corpora lutea in ovaries;
- number of implantation sites in uterus;
- number of live and dead fetuses;
- litter size;
- number of live and dead newborns;
- number of individuals of different sexes;
- weight of infants;
- time of pinna detachment;
- time of lanugo appearance;
- time of eruption of incisors;
- time of eyes opening;
- time of testes lowering;
- time of vagina opening;
- time of formation of sensory-motor reflexes.
Reproductive toxicity study according to OECD protocols on testing of chemicals:
| 414 | Prenatal toxicity |
| 415 | One-generation reproduction toxicity |
| 416 | Two-generation reproduction toxicity |
| 421 | Reproduction/Developmental Toxicity Screening Test |
| 422 | Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test |
| 443 | Extended One-Generation Reproductive Toxicity Study |